What looks alike comes together: the origin of actine's branching networks
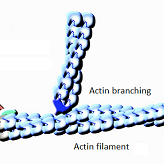

The role of the actin filaments as the skeleton of the cell is very well identified, but its implementation has so far been unclear. It is the veil of this mystery that Alexis Gautreau, CNRS Research Director at the Laboratory of Structural Biology of the Cell (BIOC)* and professor at the École polytechnique, and his team are uncovering in their latest study published today in the journal Science Advances. They show the key role of a molecular machine called dynactin, as a basis for new filaments without passing through another actin filament, thus deepening the understanding of the dynamics of the internal skeleton of cells.
> Find the publication in Science Advances
Better understanding the origin of actin networks
Dynactin plays a driving role in the movement along the microtubules, which make up the other skeleton of the cells, but despite the presence of actin-like proteins in the structure of dynactin, no role in actin polymerisation had previously been identified. Alexis Gautreau and his team have just discovered this role. They show the key role of dynactin in the formation of actin networks connected at the heart of the cell, on the surface of specific vesicles called endosomes.
The WASH molecular machine, already identified in a previous study by Alexis Gautreau's team, is responsible for the actin networks connected to the surface of the endosomes, thanks to its ability to activate the Arp2/3 machine, which also contains actin-related proteins. However, this machine only knows how to amplify filament formation by creating new filaments from pre-existing filaments. This raises the question of the first filament to initiate a connected network, and Alexis Gautreau's study reveals that this is precisely the role that dynactin fulfils. The WASH machine activates dynactin and thus coordinates two molecular machines containing actin-related proteins to initiate and amplify branched actin networks.
The work presented here therefore shows a good example of the cooperation of molecular machines involved with different polymers, actin microtubules and filaments, in endosome physiology.
* BIOC, joint research unit CNRS - École Polytechnique, Institut Polytechnique de Paris
 Support l'X
Support l'X